This is the first article in a three-part series that reviews the thermal, optical, and mechanical properties of glass. We will define common glass properties and explain their application and importance in component design.
It is critical to have a thorough understanding of glass thermal properties when designing with a glass lens or filter. When exposed to sudden or even gradual changes in temperature, improperly designed glass lenses will perform poorly and can even occasionally fail. Their thermal properties determine how they will perform in different operating conditions; this information will help you select a glass composition that will perform best for your application and environment.
Common values for the thermal properties of borosilicate glass are listed in the table below. In this article, we will discuss these properties as well as important processing temperatures.
| Thermal Property | Common Values of Borosilicate Glass |
|---|---|
| Linear Thermal Expansion | α = 30 - 60 x 10-7/°C |
| Thermal Conductivity | K = 1 Watt/m°C |
| Specific Heat | C = 800 J/kg°C |
Coefficient of Linear Thermal Expansion
The coefficient of thermal expansion (CTE) is a measure of how much volume changes as a material is heated or cooled. It is defined by
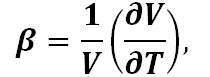
where V and T are volume and temperature, and its units are 1/°C. For glasses, the linear thermal expansion is often discussed. For isotropic amorphous materials such as glass that have small thermal expansions, the linear coefficient is accurately described by
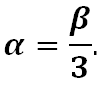
Application:
If a temperature is unevenly applied to a glass, different areas of the glass will expand by varying amounts and internal stresses will develop. This could result in glass fracture or failure.
In applications where glasses are closely installed with other materials, their thermal expansions need to match. Ceramic enamels are often applied to glass lenses to block unwanted light. The CTE for the enamel must be similar to that of the glass or the enamel will crack and chip. Another example that demonstrates the importance of CTE occurs when a glass lens is fitted tightly into a metal fixture, such as in stage lighting. If the expansions of the materials are not taken into consideration, and adequate space is not provided, then the glass could crack and fail due to applied stress from the fixture.
Thermal Shock Resistance

The thermal shock resistance of a glass indicates how likely it is to break when its temperature suddenly changes. It is defined as the maximum change in temperature (ΔT) that a glass can withstand upon rapid heating or cooling. It can be related to other glass properties by
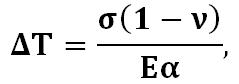
where σ is the internal stress necessary to cause cracking or failure, ν is Poisson’s ratio, E is Young’s modulus and α is the coefficient of linear thermal expansion of the glass.
Application:
Thermal shock resistance is often tested by taking heated glass lenses and rapidly cooling them through methods such as immersion in an ice bath. This type of testing can indicate the ability for glass lenses to withstand large changes in temperature when installed in application. For example, glass lenses used with high power lighting may get hot during application and experience rapid cooling when exposed to rain, snow, or other environmental factors. In these dynamic environments, it is critical to select the correct type of glass to ensure the lens’ ability to withstand thermal shock.
Thermal Conductivity
Thermal conductivity represents how well a glass conducts or transfers heat. It is defined as
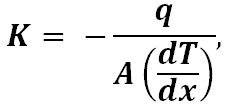
where q is the heat flow measured in watts (or J/s), A is the cross-sectional area of the glass, and dT/dx is the temperature gradient applied to the glass. Good thermal conductors will allow heat to travel through the material very quickly, much like good electrical conductors will allow for faster charge movement.
Application:
It is often desirable for glasses to have low thermal conductivity in applications and act as a thermal insulator. LEDs, for instance, perform better in cooler temperatures and will output more light, according to research performed by the Lighting Research Center. If a temperature controlled LED fixture were to operate in a hot environment, then using a glass lens with a low thermal conductivity in that fixture would reduce the heat flow through the glass to the LED and improve its energy efficiency.
Specific Heat
The specific heat of a glass is the heat needed to raise the temperature of the glass by 1°C per unit weight:
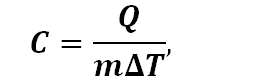
where Q is heat, m is mass and T is temperature. If the thermal conductivity shows how much heat will flow through a material, the specific heat shows how quickly heat will raise the temperature of a glass.
Application:
The specific heat of a glass part could be an important consideration for applications where the glass operates at high temperatures. Consider a lighting fixture that uses a glass lens with a quartz halogen bulb; these bulbs often operate at high temperatures, outputting a large amount of heat. If the lens is designed with a lower specific heat, it will reach its equilibrium temperature faster and reduce the warm-up time of the system.
Important Glass Temperatures
There are typically five important temperatures that are often discussed in the production and design of glasses.
- Melting point is the temperature at which the raw materials melt to a liquid state.
- Working point is the temperature at which the glass melt is shaped or molded.
- Softening dilatometric point is the temperature at which the glass begins to deform during heating when measured in a dilatometer.
- Annealing point is the temperature at which the residual stresses in a glass are reduced over a matter of minutes.
- Strain point is the temperature at which the residual stresses in a glass are reduced over a matter of hours.
These values are typically given as a range of temperatures rather than as a single point.
Application:
A sound knowledge of these temperature points is very important for glass manufacturers; it helps ensure production efficiency as well as high quality products. But it is also important for application design so that the right glass is chosen for a specific job. If a glass lens is going to be used in a high temperature environment, like the lens for a spotlight, its softening point has to be higher than the operating temperature of the light or the glass could lose its desired shape. These temperatures are also critical for setting parameters for the annealing, tempering, or heat-strengthening of glasses.
Thermal Dependence of Other Glass Properties
Many other glass properties may be affected by a change in temperature. For example, the chromaticity or color of a glass is often dependent on its thermal history. Reds and yellow colors in glass are usually developed during manufacturing by a process called striking, where the glass is reheated and cooled to promote specific colors through oxidation, reduction, or precipitation reactions of the colorants. In some cases, the glasses may even change color under normal operating conditions as the glasses equilibrate with the light source temperature.
As discussed above, glasses that are heated expand by an amount proportional to their coefficient of thermal expansion. This change in volume may also affect both the density and the refractive index of the glass. Typically, the density will decrease as the spacing between the ions in the glass increases. The refractive index, however, can either increase or decrease with temperature depending on both the change in ion spacing as well as the change in the electron cloud surrounding the ions.
Just as it is important to understand the thermal nature of a glass for temperature sensitive applications, it is often necessary to take the transmission, chromaticity, and refractive index of a glass into account during the design of a lens. The next article in this series will discuss the optical properties of glass and how these properties affect the suitability of a composition for different applications.
Learn More About Glass
To help you design better-performing glasses lenses, we created a comprehensive eBook that includes more than 40 pages of information on the thermal, optical, and mechanical properties of glass.
If you want to learn how to design glass lenses and components that are optimized for both your performance requirements and operating environment, download our free eBook.

